Research which has identified a way of making a safer and cheaper polio vaccine is to be scaled-up – to see if it could be used in commercial vaccine production.
The new approach, which has the potential to revolutionise vaccine development across the board, is the result of a scientific collaboration led by the University.
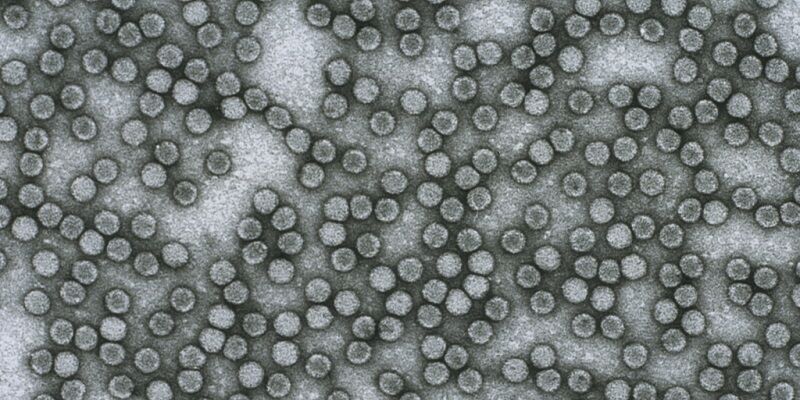
It is based on a process using harmless proteins called virus-like particles (VLPs) rather than a live virus, which carries the risk of escaping and infecting people.
CPI, an innovation "catapult" that commercialises new technologies, has created a pilot-scale manufacturing platform using the method of vaccine manufacture developed in the Leeds laboratories.
Over the next couple of months, scientists will investigate the yield and quality of vaccine produced.“This will enable the scientists at the University to optimise the process to maximise vaccine production.”
Dr Natasha Lethbridge, Senior Project Manager at CPI, said: “There are always challenges when transferring processes from the laboratory environment to an industrial manufacturing platform.
“Establishing this platform will enable the scientists at the University to optimise the process to maximise vaccine production and then establish further process scale-up models for biotherapeutic production.”
The overall aim of the project is to develop a production process that can be used by low-to-middle income countries.
The vaccine development programme has been led by Professors Nicola Stonehouse and Dave Rowlands from Leeds' Faculty of Biological Sciences.
VLPs are created in the laboratory to mimic the effects of a virus on the immune system. But they do not carry genetic material and are not infectious.
Professor Stonehouse said: “This is an exciting time for the project.
“As scientists we know that VLPs can stimulate an immune response – in effect they trick the body into thinking they are the virus, and it is that which stimulates the body’s natural immune response and gives protection from the disease.
“But VLPs can be very unstable, and that has been one of the obstacles in using them as an alternative for a live virus in vaccine manufacture. But our team has managed to overcome this problem.
“The aim now is to see if the method that we have developed in the laboratory can be scaled up and run on an industrial scale.”
Polio is a disease that causes irreversible paralysis and in some cases, death. Although it has been eradicated in large parts of the world, it still remains a significant health challenge – particularly in countries with less developed healthcare systems such as Pakistan, Afghanistan and Nigeria.
The genetic code of the poliovirus is also liable to mutation.
For these reasons, there needs to be a steady supply of vaccines to protect people. Because the new approach to the development of the vaccine uses VLPs instead of a live virus, it can be made without the need for costly bio-containment facilities.
This opens up the possibility of low-to-middle income countries developing their own production programmes, instead of having to rely on importing vaccines from other countries.
The funding for research into the use of VLPs for polio vaccine development came from the World Health Organisation. The UK Department for Health, through the Engineering and Physical Sciences Research Council, is funding the research to see if the process could be adopted by industry.
The consortium involved in the project and led by Leeds includes the Universities of Oxford, Reading and Florida, the National Institute for Biological Standards and Control, the Pirbright Institute and the John Innes Centre.
Further information
Image courtesy of Sanofi Pasteur.
For media enquiries, contact University of Leeds Media Relations and Communications Officer David Lewis via pressoffice@leeds.ac.uk.