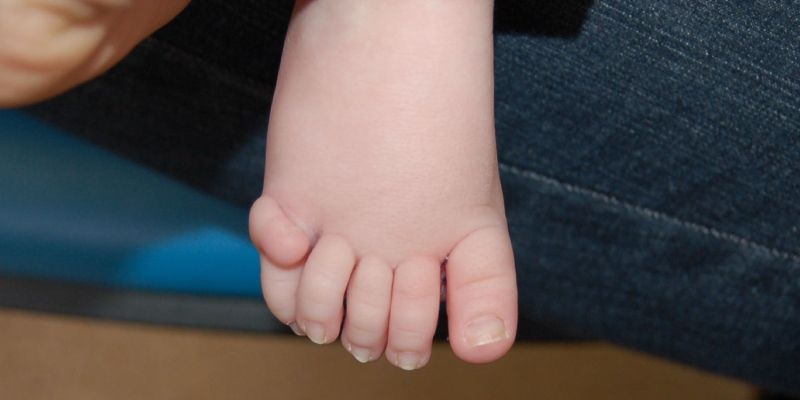
A rare disorder which causes babies to be born with extra fingers and toes and a range of birth defects has been identified in new research co-led by the University of Leeds.
The disorder, which has not yet been named, is caused by a genetic mutation in a gene called MAX. As well as extra digits – polydactyly - it leads to a range of symptoms relating to ongoing brain growth, such as autism.
The research marks the first time this genetic link has been identified. It has also found a molecule that could potentially be used to treat some of the neurological symptoms and prevent any worsening of their condition. However, more research is needed to test this molecule before it can be used as a treatment.
Published in the American Journal of Human Genetics, the paper focuses on three individuals with a rare combination of physical traits, namely polydactyly, and a much larger than average head circumference – known as macrocephaly.
The individuals share some other characteristics, including delayed development of their eyes which results in problems with their vision early in life.
It is important that these patients and their families discover the cause of their condition – and if they can access a therapy based on their genetic diagnosis, that could be life changing.
The researchers compared the DNA of these individuals and found they all carried the shared genetic mutation causing their birth defects.
The research was co-led by Dr James Poulter from the University of Leeds; Dr Pierre Lavigne at Université de Sherbrooke in Québec and Professor Helen Firth at Cambridge University.
Dr Poulter, UKRI Future Leaders Fellow and University Academic Fellow in Molecular Neuroscience, said: “Currently there are no treatments for these patients. This means that our research into rare conditions is not only important to help us understand them better, but also to identify potential ways to treat them.
“In this case, we found a drug that is already in clinical trials for another disorder – meaning we could fast track this for these patients if our research finds the drug reverses some of the effects of the mutation.
“It also means that other patients with a similar combination of features can be tested to see if they have the same variant we have identified in our study.”
Interdisciplinary research
The study team has highlighted the importance of interdisciplinary research into rare diseases in giving understanding and hope of a treatment to families who often face many years of uncertainty about their child’s condition and prognosis.
Dr Poulter added: “These are often under-represented conditions that have a huge impact on patients and their families. These families go through a long and complex diagnostic odyssey. The time from their first doctor’s visit as babies to getting a diagnosis can take more than 10 years.
“It is important that these patients and their families discover the cause of their condition – and if they can access a therapy based on their genetic diagnosis, that could be life changing.”
Dr Lavigne said: “Finding out the impact of the mutation on the function of MAX is the first step towards the development of a treatment for these children.”
The researchers now plan to look for additional patients with mutations in MAX to better understand the disorder and investigate whether the potential treatment improves the symptoms caused by the mutation.
The research was carried out in collaboration with the Leeds Teaching Hospitals Trust, the NHS Wales’ All Wales Medical Genomics Service and Radboud University Medical Center, The Netherlands.
It used data from the Deciphering Developmental Disorders study, which was led by the Wellcome Sanger Institute.
Professor Firth said: “The DDD study recruited across the UK from 2011-2015. It’s exciting that in 2024, we’re still making new discoveries. This new finding is a diagnosis for our DDD patients. Furthermore, this publication will now enable other children worldwide to be diagnosed with this novel disorder.”
Further information
“A recurrent de novo MAX p.Arg60Gln variant causes a syndromic overgrowth disorder through differential expression of c-Myc target genes” is published in American Journal of Human Genetics.
This work was funded by a UK Research & Innovation Future Leaders Fellowship to James Poulter and a Canadian Institutes of Health Research project grant (PL). The genetic sequencing was undertaken as part of the DDD (Deciphering Developmental Disorders) study which presents independent research commissioned by the Health Innovation Challenge Fund [grant number HICF-1009-003]. This study makes use of DECIPHER , which is funded by the Wellcome Trust and hosted by EMBL-EBI [grant number WT223718/Z/21/Z].